Advanced cell culture techniques form the foundation of our research in biomedical sciences and regenerative medicine. In combination with innovative materials and state-of-the-art biofabrication technologies, we establish robust platforms for translational research.
Complex tissue models as physiologically relevant alternatives to animal testing (3Rs principle) enable detailed investigation of regenerative processes and disease pathophysiology. These models are of particular relevance for basic and clinical research, as they facilitate the systematic development and preclinical assessment of novel therapeutic strategies. In this context, some of our advanced tissue models have already received great public and scientific attention: These include the Ursula M. Händel Prize 2022, which was awarded to the Würzburg Initiative 3R (WI3R) by the German Research Foundation (DFG), the Felix Wankel Prize 2021, the EPAA 3Rs Science Prize 2018 and the Lush Prize 2016. It reflects the great success of the interdisciplinary research team.
Focus areas of our research are vascularization, tumor models, musculoskeletal approaches, and biomimetic material topographies regulating cell function.
Contact:
Dr. rer. nat. Antje Appelt-Menzel
Telephone +49(0)931 31-80771
antje.appelt-menzel@uni-wuerzburg.de
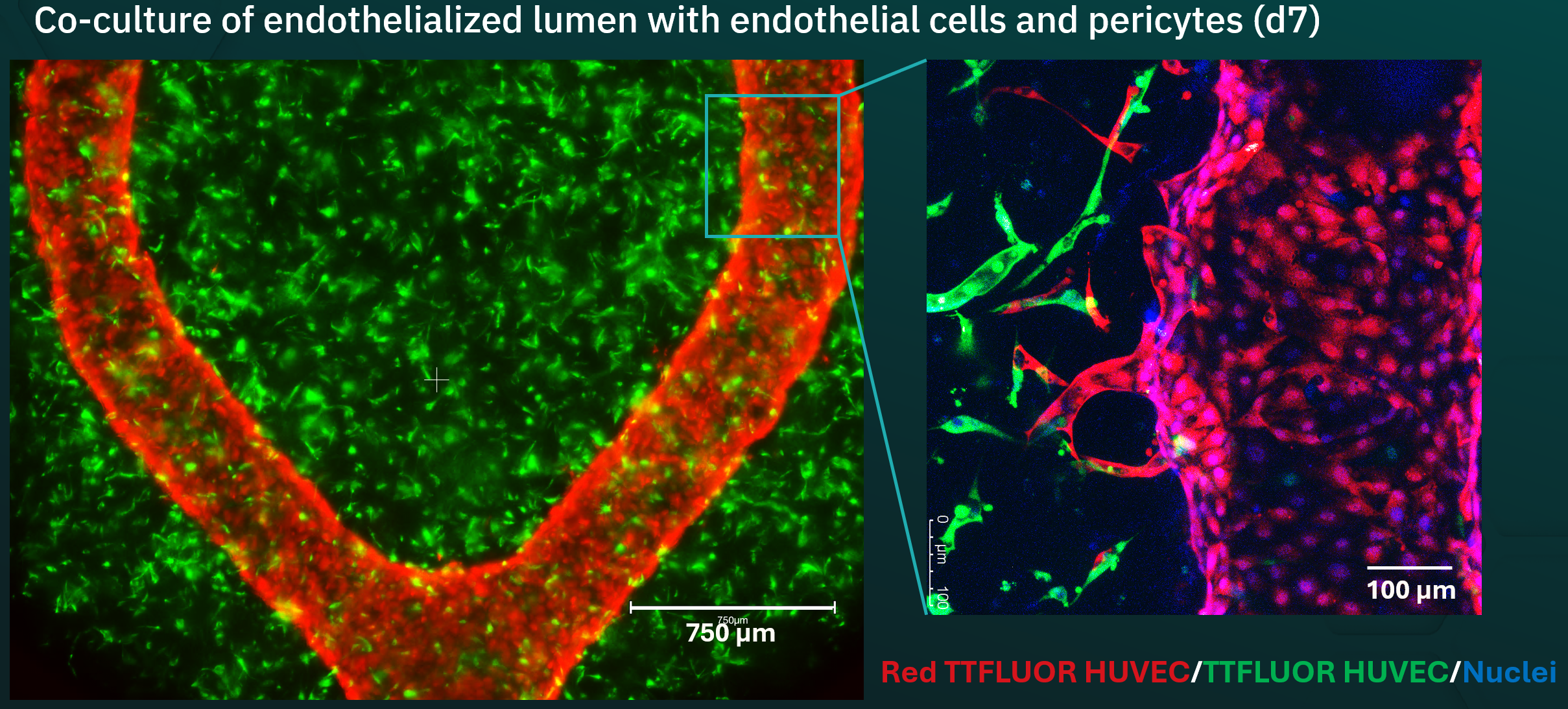
Vascularization
Vascularization is a key requirement in tissue engineering and biofabrication. Living tissues depend on vascular networks to deliver oxygen and nutrients, distribute signaling molecules, and remove metabolic waste. In engineered tissues, diffusion alone is not sufficient to support cells over larger distances. As a result, construct size and functionality are limited unless functional, perfusable vascular structures are integrated. Developing such networks is therefore essential for creating realistic and scalable tissue models.
Our platform uses innovative biofabrication strategies to enable vascularization and controlled perfusion within engineered tissues. We use volumetric bioprinting and in-gel bioprinting to create precisely defined, perfusable channels directly within cell-laden hydrogels. To mimic larger blood vessels, we fabricate artery-like structures using melt electrowriting of tubular polycaprolactone (PCL) scaffolds, providing mechanically stable templates for vascular tissue engineering. In addition, we apply sacrificial templating for microvasculature to generate interconnected channel networks that enhance nutrient supply and waste removal throughout the construct.
By combining these approaches, we aim to create viable, perfusable tissue constructs that closely resemble native tissue – advancing in vitro models and supporting future applications in regenerative medicine.
In addition to peripheral endothelial cells, we also focus on specialized endothelia within the central nervous system. A particular emphasis of our work is the blood–brain barrier and the neurovascular unit. These highly specialized structures tightly regulate the exchange between the bloodstream and the brain, protecting neural tissue while maintaining a precisely controlled microenvironment. Understanding how this barrier functions and how it can be selectively crossed is central to addressing key biomedical challenges.
Our research aims to answer critical questions such as: How can therapeutic drugs effectively enter the blood-brain barrier? How do pathogens gain access to the central nervous system? And how do disruptions of the neurovascular unit contribute to the development of neurological diseases? By unraveling these mechanisms, we seek to advance the development of improved treatment strategies for disorders of the brain and central nervous system.
Contact:
Dr. Antje Appelt-Menzel (Neurovascular Unit/blood-brain barrier)
+49(0)931 31-80771
antje.appelt-menzel@uni-wuerzburg.de
Dr. Matthias Ryma (Sacrificial templating for microvasculature)
+49(0)931 31-80415
matthias.ryma@uni-wuerzburg.de
Jun.-Prof. Dr. Tomasz Jüngst (Vascular tissue engineering)
+49(0)931 201-74081
tomasz.juengst@uni-wuerzburg.de

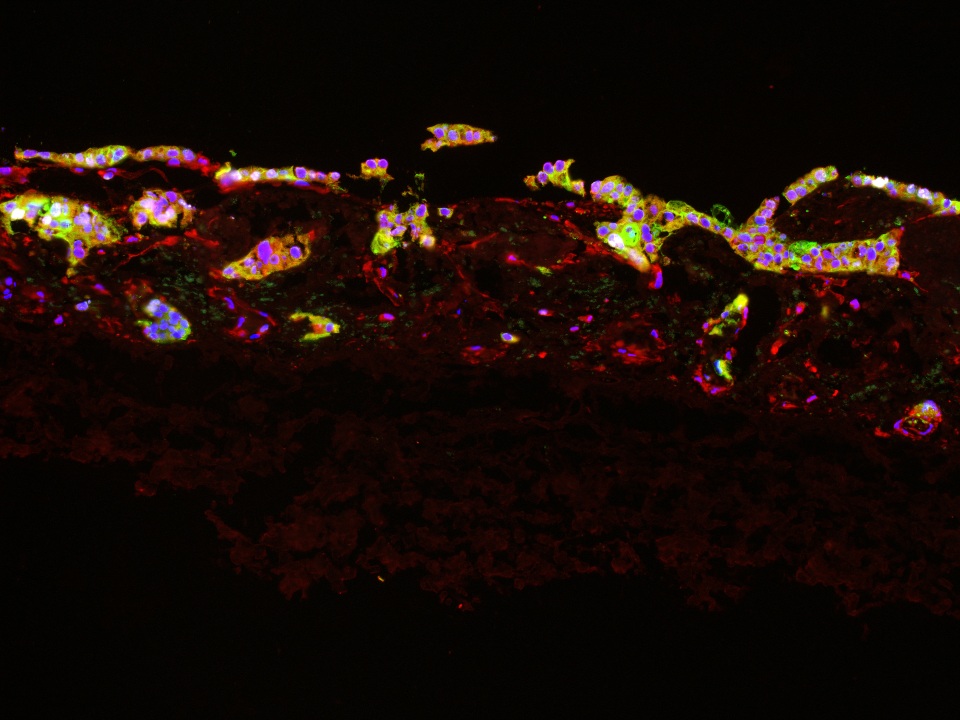
Tumor
Cancer is a complex and heterogeneous disease that remains challenging to model accurately in vitro. To better replicate tumor biology, we develop advanced 3D tumor models based on biological or synthetic matrices as well as based on organoids and spheroids. These tissue-engineered systems recreate a physiologically relevant microenvironment in which tumor (stem) cells grow within defined niche structures and exhibit clinically relevant features such as increased chemoresistance.
Our matrix-based platforms can be customized with stromal components including fibroblasts, endothelial cells, and immune cells, enabling the study of tumor – stroma interactions and immunotherapies such as CAR T cell approaches. Using both static and microfluidic cultures, we analyze immune cell infiltration and tumor cell killing in a controlled, tissue-like setting.
By applying modern fabrication techniques such as electrospinning and melt electrowriting we establish standardized, scalable, and automation-ready cancer models.
Contact:
Dr. Gudrun Dandekar
+49(0)931 31-82597
gudrun.dandekar@uni-wuerzburg.de
Dr. Sanjana Mathew-Schmitt
+49(0)931 31-89497
sanjana.mathew-schmitt@uni-wuerzburg.de
Dr. Taufiq Ahmad
+49(0)931 201-73590
taufiq.ahmad@uni-wuerzburg.de

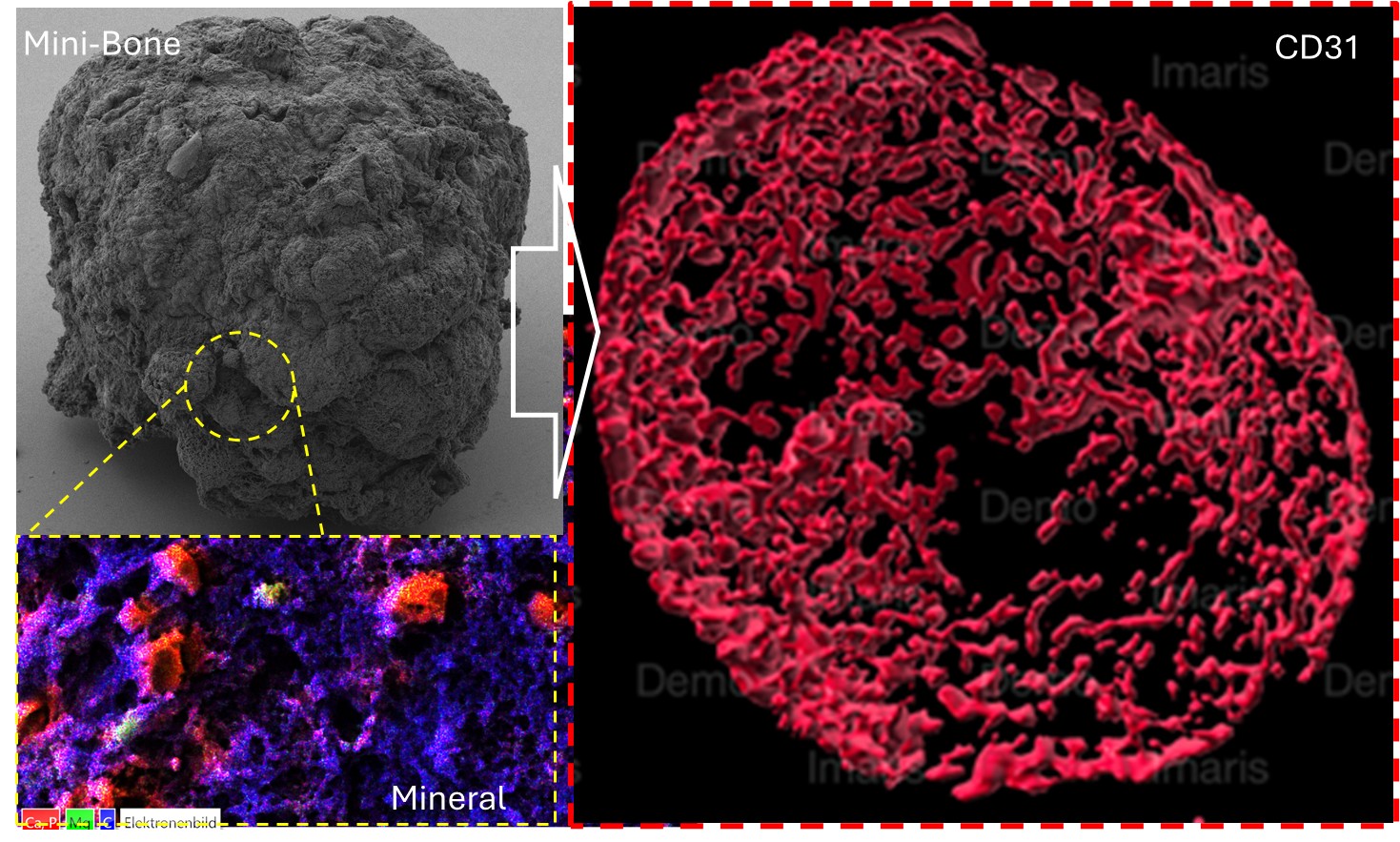
Musculoskeletal approaches
Bone Regeneration:
Bone regeneration remains a major challenge, particularly in cases of critical-size defects, impaired healing, and inflammation-associated bone loss. Our research focuses on developing advanced biomaterial and biofabrication strategies for bone and bone marrow engineering, with the aim of creating defined microenvironments that actively guide tissue formation and regeneration.
We design biomimetic scaffold systems that recapitulate key features of the native bone extracellular matrix. These platforms are based on fibrillar, collagen-inspired architectures that resemble the organic template of bone and provide instructive cues for cell adhesion, matrix organization, and osteogenic differentiation of mesenchymal stromal/stem cells (MSCs). By incorporating bioactive bioceramic particles into these scaffolds, we enable the controlled release of osteoinductive ions, which further promote bone formation while modulating inflammatory responses to support a pro-regenerative environment.
In parallel, we engineer mineralized fibrillar matrices and advanced in vitro bone models that replicate essential aspects of the natural collagen-mineral composite. These systems also allow us to recreate key features of the bone marrow niche, supporting stem cell function and enabling the development of physiologically relevant microtissues.
Using advanced fabrication technologies such as melt electrowriting and related precision processing strategies, we establish reproducible and customizable platforms for bone tissue engineering. These approaches enable the generation of mini-bone constructs, which serve both as advanced in vitro models and as modular building blocks for the fabrication of larger, bone-like tissues.
Contact:
Dr. Taufiq Ahmad
+49(0)931 201-73590
taufiq.ahmad@uni-wuerzburg.de
Dr. Kristina Andelovic
+49(0)931 201-73654
kristina.andelovic@uni-wuerzburg.de
Engineering of Tendon:
Tendon injuries and other degenerative musculoskeletal conditions remain challenging to treat, largely due to the complex, highly organized structure of these tissues, their limited blood supply, and the demanding mechanical forces they must withstand.
We develop advanced biomaterial-based platforms specifically designed for tendon regeneration. Our approach focuses on hierarchically structured, collagen-inspired nanofibrillar scaffolds that closely mimic key features of the native tendon extracellular matrix. These include aligned nanofiber architectures and biomimetic surface patterns, such as subtle, wave-like microstructures, that help guide cell behavior, support organized tissue formation, and enhance mechanical performance.
To further improve durability under mechanical load, we incorporate elastic, polycaprolactone-based materials alongside tailored fiber designs. This combination increases flexibility, toughness, and resistance to repeated stress.
In combination with mesenchymal stem cells, these bioinspired systems enable us to study tendon-specific cell differentiation, matrix formation, and the development of functional, tendon-like tissue in a mechanically relevant environment.
By leveraging anisotropic, compliant fibrillar structures, our work promotes cell alignment, efficient force transmission, and robust tissue maturation – laying the foundation for systematic studies of cell-material interactions and the development of next-generation regenerative therapies.
Contact:
Dr. Kristina Andelovic
+49(0)931 201-73654
kristina.andelovic@uni-wuerzburg.de
Muscle Tissue Engineering:
Biofabrication strategies for skeletal muscle tissue engineering are another strong research focus. We design cell-instructive scaffolds with controlled architecture and topography to support stem cell alignment, myogenic differentiation, and the formation of organized muscle-like tissues. These platforms help recreate essential features of native muscle in a controlled in vitro setting.
Contact:
Dr. Taufiq Ahmad
+49(0)931 201-73590
taufiq.ahmad@uni-wuerzburg.de
Interface Scaffold Design:
Our research also extends to the development of advanced scaffold systems for musculoskeletal interfaces, bridging the gap between our work on bone, muscle and tendon regeneration. We focus on complex transition zones such as bone-tendon and muscle-tendon interfaces, where gradients in structure, composition, and mechanics are essential for proper function. By engineering scaffold microgeometry, surface properties, and material gradients, we aim to recreate the hierarchical organization and biological complexity of these native interfaces. These integrative platforms enable a deeper understanding of tissue connectivity and support the development of more effective regenerative strategies for multi-tissue systems.
Contact:
Dr. Taufiq Ahmad
+49(0)931 201-73590
taufiq.ahmad@uni-wuerzburg.de
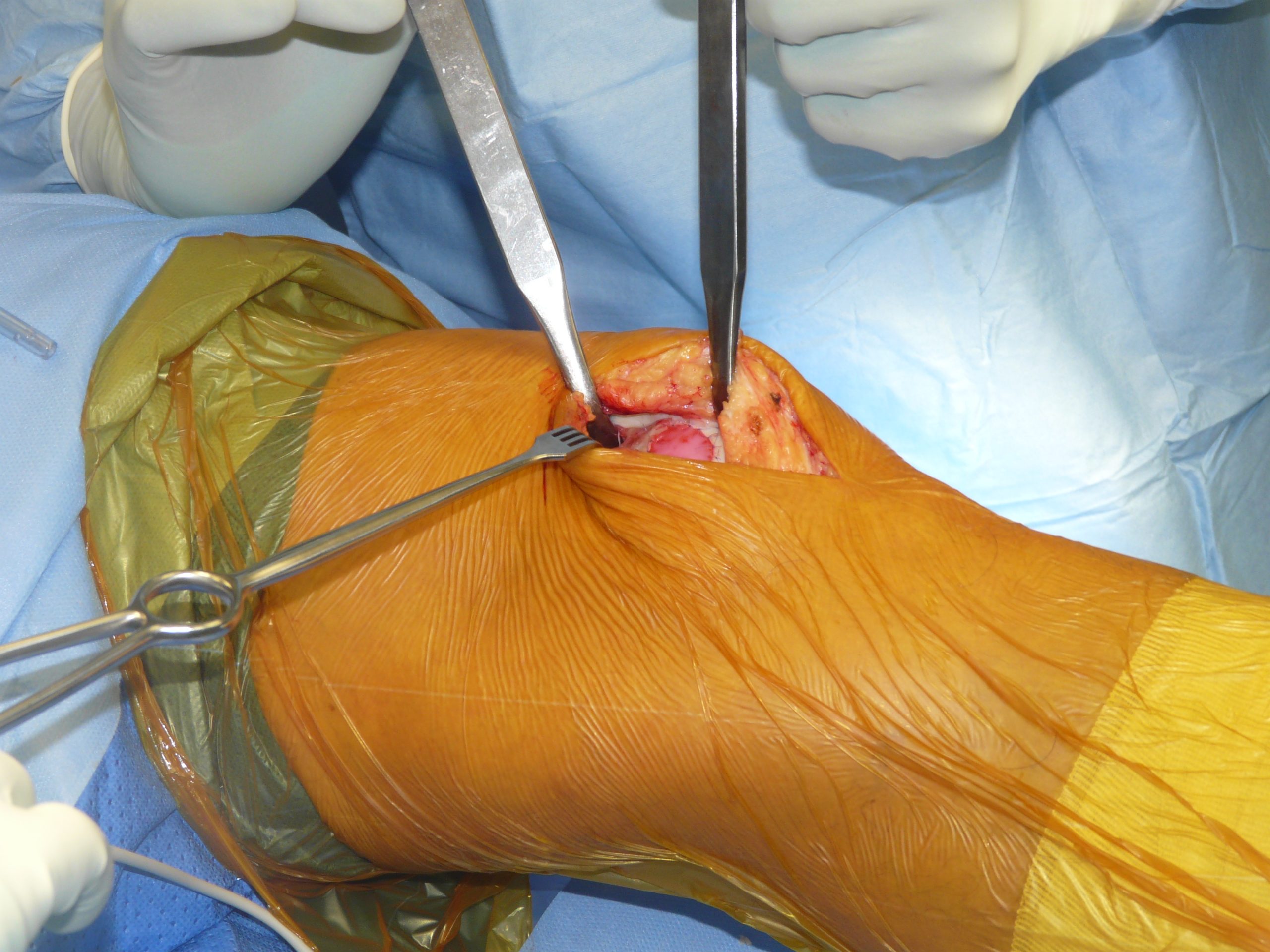
Cartilage – GMP compliant ATMP production:
Articular cartilage of the knee has only a very limited intrinsic capacity for regeneration. Once damaged by trauma or degenerative conditions such as Osteoarthritis, defects typically progress over time – leading to chronic pain, reduced mobility, and ultimately the need for joint replacement surgery. GMP-compliant, tissue-engineered cartilage implants offer a biologically driven alternative. Rather than merely alleviating symptoms or replacing the entire joint, this approach aims to restore joint function by repairing defects with living, patient-specific tissue in a personalized medical approach.
Cartilage production is currently centered on nasal cartilage-derived, tissue-engineered implants, which are manufactured at University Hospital Würzburg. The product is classified as an Advanced Therapy Medicinal Product (ATMP) and is being evaluated in 2 phase II clinical trials. The process involves harvesting cartilage cells from the nasal septum, expanding them in vitro, and cultivating them on a collagen scaffold to form functional cartilage tissue. The engineered constructs mature for approximately four weeks in state-of-the-art clean rooms at the University Hospital Würzburg GMP Facility. Production adheres strictly to Good Manufacturing Practice (GMP) standards applicable to medicinal products and medical devices.
This approach represents an innovative strategy in orthopedic regenerative medicine, aiming to restore joint function and improve quality of life.
Contact:
PD Dr. Oliver Pullig
+49(0)931 31-83748
oliver.pullig@uni-wuerzburg.de
Dr. Sarah Nietzer
+49(0)931 31-82596
sarah.nietzer@uni-wuerzburg.de
Sebastian Häusner
+49(0)931 31-87955
sebastian.haeusner@uni-wuerzburg.de

Biomimetic material topographies regulating cell function
Stem cell differentiation, Immune modulation
Dr. rer. nat. Antje Appelt-Menzel
Telephone +49(0)931 31-80771
antje.appelt-menzel@uni-wuerzburg.de
Geraldine Beer
Sebastian Beusink
Sabina Eibichova
Markus Glaser
Melanie Hein
Julian Schwebler
Fabiola Walz
Camilla Mussoni
Muhammad Aslam
Dr. Gudrun Dandekar
Dr. Sanjana Mathew-Schmitt
Dr. Kristina Andelovic
Dr. Taufiq Ahmad
PD Dr. Oliver Pullig
Dr. Sarah Nietzer
Sebastian Häusner
Dr. Matthias Ryma
Dr. Thorsten Keller
Dr. Christian Lotz
Dr. Maximiliane Wußmann
Vascularization
Tumor models
Musculoskeletal approaches
Heinze, C., Mussoni, C., Ryma, M., Gergely, C., Lamberger, Z., Schäfer, A., Andelovic, K., Stahlhut, P., Lang, G., Groll, J. and Ahmad, T., 2025. Bioassembly of Myoblast Spheroids in Electrofibrillated Scaffolds for 3D Muscle Tissue Biofabrication. Small, 21(41), p.e03351.
Mussoni C, Heinze C, Ryma M, Jun I, Lamberger Z, Andelovic K, Kade JC, Stahlhut P, Lang G, Ahmad T, Groll J. Muscle-fiber-inspired nanofibrillar microbundles induce myogenic differentiation in human adipose-derived stem cells. Bioactive Materials. 2026 Sep 1;63:319-35.
Wixmerten A, Miot S, Bittorf P, Wolf F, Feliciano S, Hackenberg S, Häusner S, Krenger W, Haug M, Martin I, Pullig O, Barbero A. Good Manufacturing Practice-compliant change of raw material in the manufacturing process of a clinically used advanced therapy medicinal product-a comparability study. Cytotherapy. 2023 May;25(5):548-558 Epub 2023 Mar 7. PMID: 36894437
Haeusner S, Herbst L, Bittorf P, Schwarz T, Henze C, Mauermann M, Ochs J, Schmitt R, Blache U, Wixmerten A, Miot S, Martin I, Pullig O. From Single Batch to Mass Production-Automated Platform Design Concept for a Phase II Clinical Trial Tissue Engineered Cartilage Product. Front Med (Lausanne). 2021 Aug 13;8:712917 PMID: 34485343; PMCID: PMC8414576
- From 2025 to 2028, we are part of the BMFTR-funded research project KITAMAKI (03VP12662). Together with the University of Würzburg (Immunology) and the Translational Center at Fraunhofer ISC, we are developing and evaluating an innovative antibody-coated fleece designed to locally activate resident immune cells in breast cancer skin metastases.(https://www.validierungsfoerderung.de/validierungsprojekte/kitamaki and https://www.med.uni-wuerzburg.de/aktuelles/meldungen/single/news/kitamaki/)
- We are part of the DFG funded Collaborative Research Centre/Transregio CRC/TRR 225, “From the Fundamentals of Biofabrication towards Functional Tissue Models,” uniting the Universities of Würzburg, Bayreuth, and Erlangen–Nürnberg. In subproject C07, we focus on vascularization as a key challenge in biofabrication. Using advanced free-form printing of POx-based sacrificial materials, we create complex, perfusable microvascular networks and integrate stable vessel-like structures into 3D tissue models. Our aim is to develop standardized in vitro systems that better replicate physiological conditions and improve predictive power in preclinical research.
(Project C07 – SFB TRR 225 biofab) - Since 2022, Dr. Ahmad is leading the DFG-funded CRC/TRR 225 subproject C06, “Biofabrication strategies for modelling kidney compartments and diseases,” together with Prof. Dr. Janina Müller-Deile. The project develops advanced in vitro kidney models to better reproduce native renal complexity and reduce reliance on animal experiments. Current work includes dynamic models of the glomerular filtration barrier based on electrospun biomimetic scaffolds and bioreactor culture, as well as next-generation proximal tubule and vascularized organoid-based systems for studying inflammation, fibrosis, and drug responses in patient-relevant settings.
(Project C06 – SFB TRR 225 Biofab). - Horizon EIC Transition project Vasc-on-Demand (Project No. 101156395) www.vasc-on-demand.eu
- IZKF Projekt T-535 (Herrmanns, Ryma): Biofabrikation vaskularisierter Lebermodelle: Progression primärer Langzeit-Hepatozytenkulturen
https://www.med.uni-wuerzburg.de/izkf/forschungsfoerderung/aktuell-laufende-izkf-foerderungen/207871/ - We are part of ENCANTO, a Horizon Europe–funded health project running from 2024–2029. The project centers on a regenerative cell-therapy approach where cartilage grafts are engineered from nasal cartilage cells to repair damaged cartilage under the kneecap in patellofemoral (kneecap) osteoarthritis. The multicentric clinical trials are conducted in 11 clinical centers across Europe, with production sites in Basel and Würzburg.
https://encanto.health/ - We are partner in IICT-PFOA2, an investigator initiated clinical trials founded by the Swiss National Foundation. Together with leading hospitals in Switzerland, Germany, and Croatia, we produce our advanced tissue-based medicinal product N-TEC (nasal chondrocyte tissue-engineered cartilage) for the treatment of patellofemoral osteoarthritis.
https://data.snf.ch/grants/grant/214562 - We are part of LUMINATE, a Horizon Europe funded project running from 2024-2029. The project aims to combine filament light bioprinting, micro‑extrusion, innovative biomaterials, and regenerative medicine to repair both cartilage and the underlying subchondral bone in situ in a one-step approach.
https://luminateproject.eu/ - In the EU COST action NetwOArk, Dr. Oliver Pullig is member of the managment committee for Germany and involved in WG2 “Speeding up translation from bench to bedside” and in WG 4 “Building the European Society for Osteoarthritis (NetwOArk)”. The main aim of the netwOArk is to involve the three major stakeholder groups: Patients, clinicians and researchers in the field of osteoarthritis.
https://netwoark.eu
